8 min
Medically reviewed: • Sources verified:Retatrutide Phase 2 Results (2025): Weight Loss Data, Safety, and Next Steps
Uncover retatrutide phase 2 results: up to 24.2% weight loss at 48 weeks, HbA1c reductions in T2D, mild GI side effects, and phase 3 progress toward FDA approval. Full efficacy, safety breakdown.
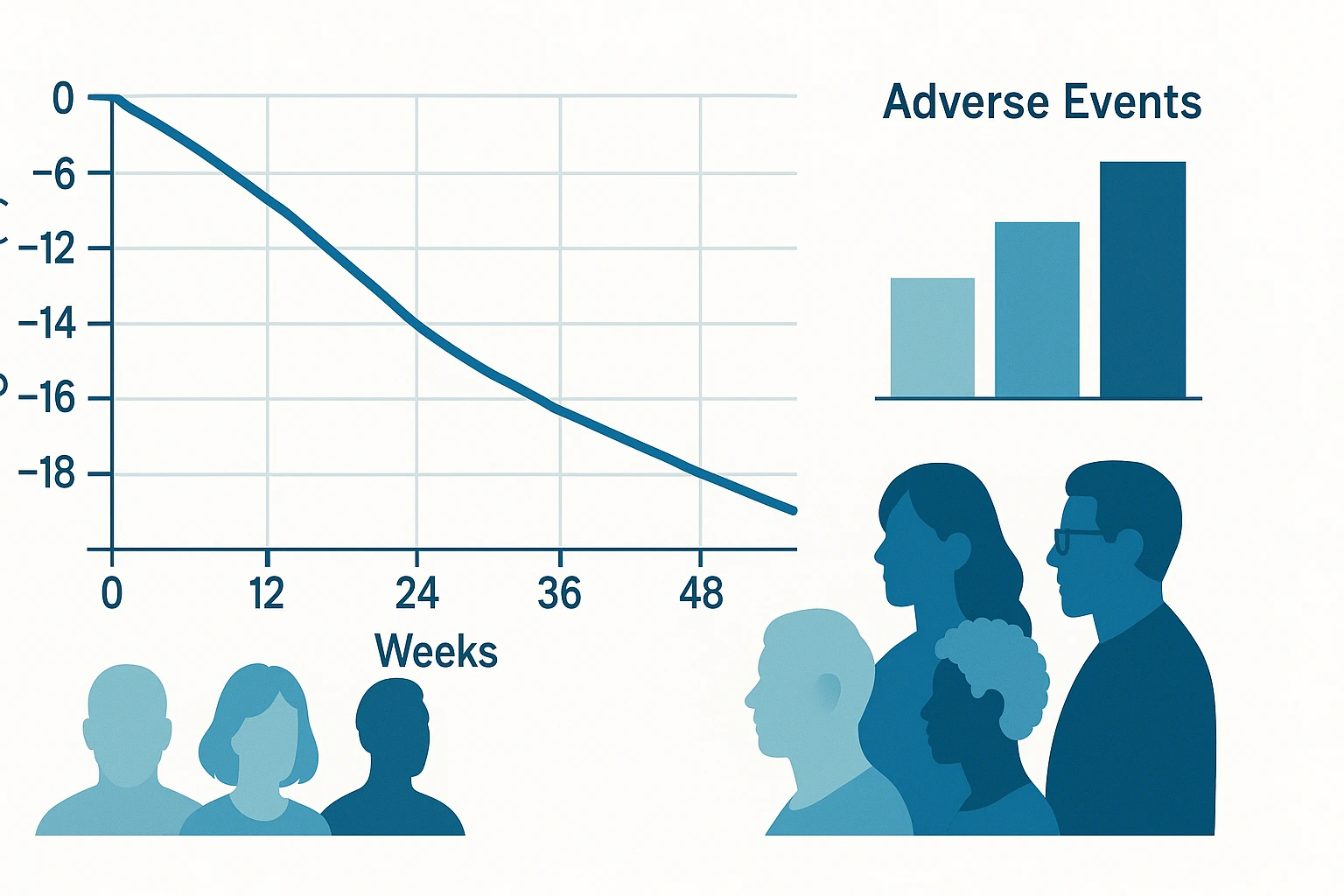
Retatrutide phase 2 results revealed impressive weight loss of up to 24.2% at 48 weeks with the 12 mg dose, surpassing many current treatments for obesity [1]. The trials also showed strong improvements in blood sugar control for people with type 2 diabetes (T2D) and better heart health markers [1][3]. These findings from Eli Lilly's studies pave the way for phase 3 trials, though safety concerns like stomach side effects need monitoring [1]. For current retatrutide FDA approval details, see our dedicated guide.
Introduction to Retatrutide Phase 2 Results
Phase 2 trial data from key clinical studies tested this new drug for obesity and related conditions. Published in top medical journals, these outcomes highlight its potential as a game-changer [1].
Key Highlights from the Trials
- Highest dose (12 mg) led to 24.2% average weight loss at 48 weeks, with no signs of plateauing [1].
- Nearly all participants (100%) on 8 mg or 12 mg doses lost at least 5% body weight; 93% lost 10% or more [1].
- Glycemic control improved dramatically in T2D patients, with HbA1c drops of 1.3-2.0% [3].
These study outcomes build excitement for better obesity treatments.
Why These Results Matter for Obesity and T2D Treatment
Obesity affects millions, raising risks for diabetes, heart disease, and more. These phase 2 findings show deeper weight loss than drugs like semaglutide or tirzepatide in early tests [1].
For T2D, achieving HbA1c below 6.5% in up to 82% of patients could mean fewer complications [3]. Overall, the outcomes suggest retatrutide could fill gaps in current therapies.
What is Retatrutide?
Retatrutide, also known as LY3437943, is an experimental drug from Eli Lilly. It targets obesity, overweight, T2D, and liver fat issues like NAFLD [1][2].
Mechanism of Action: Triple Agonist (GLP-1, GIP, Glucagon)
Unlike single or dual agonists like Ozempic or Mounjaro, retatrutide activates three key receptors [1]:
- GLP-1: Reduces appetite and slows digestion.
- GIP: Boosts insulin and aids fat metabolism.
- Glucagon: Increases energy use and burns fat.
This triple action explains the strong phase 2 results in weight loss and metabolism.
Developer: Eli Lilly and Trial Overview (NCT04881760)
Eli Lilly developed retatrutide for weekly injections. The main phase 2 trial NCT04881760 included 338 adults with obesity or overweight (no T2D initially) [2].
Subgroups tested T2D and NAFLD effects [1][3]. Results supported moving to larger phase 3 studies.
Phase 2 Trial Design and Doses Tested
The trials were randomized, double-blind, and placebo-controlled for reliable data [2]. They lasted up to 48 weeks to check long-term effects [1].
Study Population: Obesity, Overweight, T2D, and NAFLD Subsets
- Main group: Adults with BMI ≥30 (obese) or ≥27 (overweight) with issues like high blood pressure [2].
- Subsets: People with T2D or NAFLD for targeted data [1][3].
- Total: Around 338 in obesity trial; others in related studies [2].
This diverse group mirrors real-world patients.
Dosing Regimen: 1mg, 4mg, 8mg, 12mg and Dose Escalation
Doses were weekly subcutaneous shots with slow escalation to reduce side effects [1][2]:
| Dose | Escalation Start | Maintenance |
|---|---|---|
| 1 mg | Low, steady | 1 mg weekly |
| 4 mg | From 2 mg | 4 mg weekly |
| 8 mg | From 2-4 mg | 8 mg weekly |
| 12 mg | From 2 mg | 12 mg weekly |
Placebo group got dummy shots [2].
Endpoints: 24 Weeks (Primary) and 48 Weeks (Secondary)
- Primary: Percent weight change at 24 weeks [2].
- Secondary: Weight at 48 weeks, HbA1c, lipids, safety [1].
- Responders: Categories like ≥15% loss [1].
Retatrutide Phase 2 Efficacy: Weight Loss Results
The retatrutide phase 2 results showcased impressive weight loss efficacy, with outcomes that were dose-dependent and sustained. Higher doses beat placebo by wide margins [1].
24-Week Weight Reduction by Dose (LS Mean % Change)
| Dose | % Change | Placebo-Adjusted |
|---|---|---|
| 1 mg | -7.2% | -5.6% |
| 4 mg | -12.9% | -11.3% |
| 8 mg | -17.3% | -15.7% |
| 12 mg | -17.5% | -16.0% |
| Placebo | -1.6% | - |
Absolute loss at 12 mg: 41 lbs (18.7 kg) [1][2].
48-Week Weight Loss: Up to 24.2% at 12mg (No Plateau)
Weight kept dropping through 48 weeks [1]:
| Dose | % Change | Placebo-Adjusted | Absolute Loss (12 mg example) |
|---|---|---|---|
| 1 mg | -8.7% | -6.6% | - |
| 4 mg | -17.1% | -15.0% | - |
| 8 mg | -22.8% | -20.7% | - |
| 12 mg | -24.2% | -22.1% | 58 lbs / 26 kg |
| Placebo | -2.1% | - | - |
No plateau means ongoing benefits [1].
Weight Loss Milestones: ≥5%, ≥10%, ≥15% Responders
At 48 weeks (12 mg dose) [1]:
- ≥5%: 100% of participants.
- ≥10%: 93%.
- ≥15%: 83%.
Even 4 mg hit 92%/75%/60%. High responder rates stand out.
Absolute Weight Loss (lbs/kg) and Placebo Comparison
A 200-lb person on 12 mg might lose 48 lbs by 48 weeks. Placebo lost just 4 lbs on average. Superior to many approved drugs [1].
Glycemic Control and T2D Outcomes in Phase 2
In T2D subsets, these findings improved blood sugar beyond weight loss alone [1][3].
HbA1c Reductions: 1.3-2.0% Across Doses
Over 24-36 weeks [3]:
- 4 mg: -1.3%.
- 8-12 mg: Up to -2.0%.
Big drops for uncontrolled T2D.
Achievement Rates: Up to 82% <6.5% HbA1c, 31% <5.7%
- <6.5% (target): 82% on higher doses [1].
- <5.7% (normal): 31% [1].
Fasting glucose also fell sharply.
Comparison to Dulaglutide
Retatrutide beat dulaglutide 1.5 mg (a GLP-1 drug) in HbA1c and weight [1]. Dual benefits for T2D patients.
Cardiometabolic Improvements from Retatrutide Phase 2
Retatrutide phase 2 results extended beyond weight loss to boost heart and metabolic health markers [1].
Waist Circumference, Blood Pressure, Lipids (Triglycerides, Non-HDL)
- Waist: Reduced by 7-10 inches on higher doses [1].
- Blood pressure: Systolic drops of 5-8 mmHg [1].
- Lipids: Triglycerides down 25-30%; non-HDL cholesterol improved [1].
These cut cardiovascular risks.
NAFLD Subset Benefits and No Hepatotoxicity Signals
In NAFLD patients, weight loss matched others; liver enzymes stayed stable or improved. No liver safety issues at 48 weeks [1].
Safety Profile and Side Effects in Retatrutide Phase 2 Results
Phase 2 outcomes showed a safety profile like approved GLP-1 drugs. Most issues were manageable [1].
Adverse Events Incidence: 73-94%, Mostly Mild-Moderate GI
73-94% reported events, rising with dose. But serious ones were rare [1].
Common Side Effects: Nausea, Vomiting, Diarrhea During Escalation
- Nausea: 40-60%, peaked early [1].
- Vomiting/Diarrhea: 20-40% [1].
- Mostly during ramp-up; faded over time.
Heart Rate Changes: Dose-Dependent, Peaked at 24 Weeks
Increases of 5-10 bpm at higher doses, max at week 24, then dropped. Monitored closely [1].
Discontinuations (6-16%) and Serious AEs (0-6%)
- Dropouts: 6% (1 mg) to 16% (12 mg) vs. 0% placebo [1].
- Serious AEs: 0-6% vs. 4% placebo [1].
No deaths or major concerns.
Mitigation Strategies and Comparison to GLP-1 Agonists
Slow escalation and lower starts cut GI issues. Safety matches semaglutide/tirzepatide [1]. For retatrutide's legal status in the US post-Phase 2, note it's research-only now.
Publication and Clinical Trial Status Post-Phase 2
These findings gained credibility through peer review [1].
NEJM Publication (PMID: 37366315)
Detailed in NEJM (2023) [1]. Confirms efficacy/safety data.
Phase 3 TRIUMPH Program: Ongoing Confirmation Trials
TRIUMPH phase 3 tests larger groups with active comparators [4]. Track retatrutide approval status with FDA, EMA, TGA updates for live progress.
Regulatory Status and Path to FDA Approval
Phase 2 success advanced retatrutide, but approval needs phase 3 data [1][4].
Current Legal and FDA Status After Phase 2
Not FDA-approved; investigational only. Available via trials/compassionate use? Check Is retatrutide legal in the US right now?.
What Phase 2 Results Mean for Future Approval
Strong efficacy/safety supports fast-track potential. Phase 3 to confirm; approval possible by 2026-2027 if successful [1][4]. See Is Retatrutide FDA approved? for Phase 2's role in timelines.
Conclusion: Future of Retatrutide Based on Phase 2 Data
The retatrutide phase 2 results promise up to 24% weight loss, T2D control, and cardiometabolic gains with tolerable safety [1].
Summary of Efficacy, Safety, and Next Steps
- Efficacy: Best-in-class weight loss, no plateau [1].
- Safety: GI-focused, like peers; monitor heart rate [1].
- Next: Phase 3 TRIUMPH readout soon [4]. Retatrutide approval tracker for latest FDA/EMA progress.
Potential Impact on Weight Loss Treatments
These phase 2 findings position retatrutide to outperform current leaders in obesity care. Semaglutide typically yields 15-17% weight loss, while tirzepatide hits 20-22% in head-to-head trials—yet retatrutide reached 24.2% with sustained progress and no plateau [1]. This could help more patients hit clinically meaningful targets like 15-20% loss, reducing risks for T2D, heart disease, and NAFLD more effectively.
If phase 3 confirms these retatrutide phase 2 results, it may redefine standards, offering a potent weekly option alongside lifestyle changes. Competition could drive down costs and improve access for millions affected by obesity. Patients and doctors should watch developments closely—consult a healthcare provider for personalized advice. See tirzepatide vs. retatrutide for side-by-side comparisons.
References
-
Jastreboff AM, Aronne LJ, Ahmad NN, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity - A Phase 2 Trial. N Engl J Med. 2023;389(6):514-526. doi:10.1056/NEJMoa2301972. Full text; PMID: 37366315.
-
ClinicalTrials.gov. NCT04881760: Retatrutide (LY3437943) in Participants Who Have Obesity or Overweight With Weight-Related Comorbidities Without Diabetes. U.S. National Library of Medicine. Trial record.
-
ClinicalTrials.gov. NCT04867785: Retatrutide (LY3437943) in Participants With Type 2 Diabetes (OBESE). U.S. National Library of Medicine. Trial record.
-
ClinicalTrials.gov. NCT05929066: A Study of Retatrutide (LY3437943) in Participants Who Have Obesity or Overweight With Weight-Related Comorbidities Without Diabetes (TRIUMPH-1). U.S. National Library of Medicine. Phase 3 trial record.
Related Articles
- /articles/is-retatrutide-fda-approved – FDA status update.
- /articles/retatrutide-approval-tracker-fda-ema-tga – Global trackers.
- /articles/is-retatrutide-legal-in-us – US legality.
- /articles/semaglutide-vs-retatrutide – Comparisons.
- /articles/tirzepatide-vs-retatrutide – Dual vs. triple agonists.
Sourcing research‑grade retatrutide?
Compare verified research peptide vendors, review COAs, and evaluate pricing with our comprehensive buyer's guide. All materials are intended strictly for in‑vitro laboratory research.
Ready to explore medical weight management?
Consult with US-based telehealth providers to discuss FDA-approved GLP-1 medications and personalized obesity treatment plans.